Definition: malignant neoplasm of the skin arising from the basal layer of the epidermis
Incidence/ Prevalence: Basal cell carcinoma accounts for about 90% of eyelid malignancies and about 80% of skin malignancies. Over 1 million cases of basal cell carcinoma occur in the U.S. each year and many of them are on the face, well within the domain of the ophthalmologist.
Etiology: As the name implies, basal cell carcinomas presumably originate from the stratum basale, or stratum germinativum, of the epidermis. UV exposure (especially episodic) is a key risk factor for the tumor. There is evidence of abnormalities in the sonic hedgehog signalling pathway. Sonic hedgehog protein (SHH) binds to a transmembrane tumor suppressor protein abbreviated PTCH1. This binding prevents the normal interaction with another transmembrane protein (SMO). SMO interacts through the GL1 family of transcription factors (Reference 1 below).
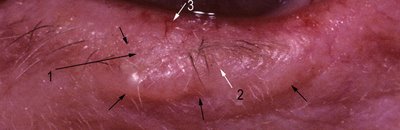 Clinical Findings:
Clinical Findings: The classic description is a “rodent” ulcer—that is a slowly enlarging ulcer with pearly, raised, rolled edges. In the photograph of the eyelid basal cell carcinoma the borders are clearly raised (short arrows) and a multinodular contour is evident. There is loss of eyelid cilia focally (arrows 1 and 2) associated with a depression in the center of the lesion (white arrow 2). There are dilated vessels at the eyelid margin (white arrow 3). Morpheaform or fibrosing basal cell carcinoma presents as a flat or slightly depressed pale yellow indurated plaque. Lesions typically occur on the face, and the lower eyelids are more commonly involved than the upper eyelids. Tumors in the medial canthal area are more likely to be deeply invasive and to involve the orbit.
Histopathology: The characteristic features and criteria for the diagnosis of basal cell carcinoma include
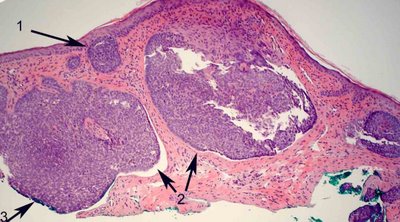
the nodular architecture conferred by the tumor and nests of basaloid cells that extend in contiguity from the basal layer of the epidermis and have a similar appearance to the stratum basale (arrow 1 above).
The nests and strands exhibit peripheral palisading. Usually single cells ring the nests with their long axis oriented radially from the center of the nest creating a picket fence arrangement. Black arrow 2 above and below.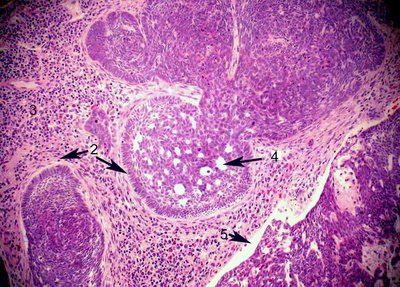
Tumor nests exhibit retraction artifact where there is a cleft created from processing between the nests and the surrounding stroma (Arrows 2 above, and 2 & 5 below).
Cytologic features include the crowding of tumor cells, which exhibit nuclear enlargement and hyperchromasia with a variable amount of atypia. In some tumors the cytologic changes may seem somewhat bland. Anaplastic features are uncommon. Basal cell carcinomas may exhibit a variety of cytologic and architectural patterns, including morpheaform, squamous, sebaceous, adenoid (arrow 4 above), and cystic differentiation. The morpheaform, or fibrosing, basal cell carcinoma, is often infiltrative, and its extent is difficult to determine clinically. Inflammation often accompanies basal cell carcinoma (number 3 above, no arrow).
Treatment: Complete excision is considered the treatment of choice. Notice in the first image above there is green ink at the edge of the specimen indicating that the margin is positive, (arrow number 3) If the lesion is characteristic clinically, many clinicians will attempt a complete excision on the first operation while others prefer a small biopsy followed by complete excision. Some will attempt to use frozen section and Moh’s procedures if the lesion involves the medial canthus. In our referral practice we have seen frozen sections of skin misinterpreted. For example the hair follicle apparatus is frequently misinterpreted as basal cell carcinoma. This is generally due to the difficulty presented in getting high quality sections from frozen skin. We advocate using a rapid turnaround with paraffin embedding for reconstruction if the initial excision is not complete. That being said, one should remember that only about 30-40% of basal cell carcinomas recur even when the margins of excision are positive.
Prognosis: Morbidity in basal cell carcinomas is almost always the result of local spread; metastatic spread is extremely unusual. The early lesion is curable and quite simple to extirpate but often missed by the less observant.
Reference:
Rubin AI et al. NEJM2005; 353:2262-2269.