Definition: Infection of the cornea caused by fungus.
Incidence/Prevalence: The incidence is less than 5% of corneal infections in the U.S. The most common organisms include Aspergillus, Candida, and Fusarium. There is a predilection for fungal keratitis for warmer humid climates. Aspergillus and Fusarium are more common in the southern U.S. Candida makes up a greater percentage of fungal keratitis in the northern U.S.
Etiology: Plant matter bearing fungus that penetrates the cornea (e.g. gardening) is a common source of fungal keratitis. Contact lens wear, use of topical steroids, cornea surgery, or keratitis from non-fungal causes are all risk factors. Candida is the most common cause of fungal keratitis. Paecilomyces lilacinus is known to contaminate intraocular lenses. Aspergillus is also a common source of fungal keratitis.
Clinical Findings: Patients may present with a dry fluffy infiltrate in the cornea with feathered margins that are usually white to gray in color. The lesions are usually slowly expansive and indolent and as it progresses to involve the deeper stroma.

In the image one can compare the region of necrotizing keratitis (yellow arrow 1) to the relatively less involved cornea (yellow arrow 2) to see the dense white infiltrate that opacifies the cornea. The fungus has infiltrated and destroyed much of the deep stroma to involve the anterior chamber (#
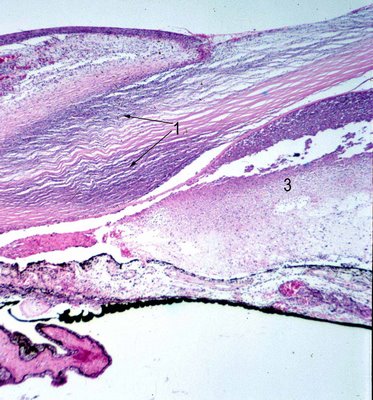
3 black arrow). A histologic section in the same area is shown below (black arrows). The initial signs of inflammation may be minimal but as the disease worsens so does the ocular injection.
 Histopathology:
Histopathology: The hallmark of fungal keratitis is the necrotizing keratitis (black arrow 1) that in general is associated with both an acute and granulomatous inflammatory infiltrate. Neutrophils (#3 black) as well as histiocytes and even multinucleated giant cells comprise the infiltrate. In the cornea there is often ulceration and a severe necrotizing stromal keratitis. In this setting special stains such as Grocott-Gomori methenamine-silver nitrate (GMS stained image #5),

and periodic acid Schiff will often highlight the fungal elements.
Fungus has penetrated to the anterior chamber (black arrow 5).
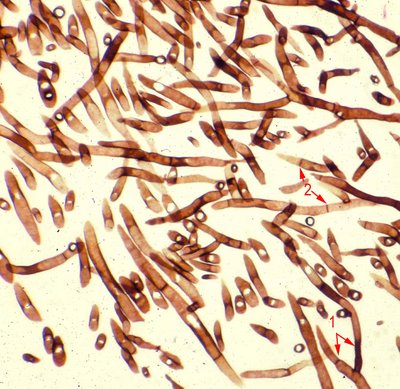
The high magnification image shows a fungus with hyphal structures that branch into 2 parts (dichotomous) at 45 degree angles (arrow 1). The hyphae are 'sepatated'.
This is a section of Aspergillus.
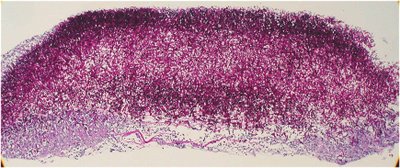
In the image shown above, there is a large fungal mass that has infiltrated and replaced the necrotic cornea. Descemet's membrane may be seen at the bottom of this PAS stained slide.
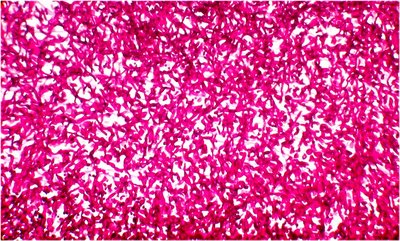
At higher magnification once sees a mass of pseudohyphae that are so dense that the branching pattern is difficult to discern but the fungal elements are clearly very strongly PAS positive.
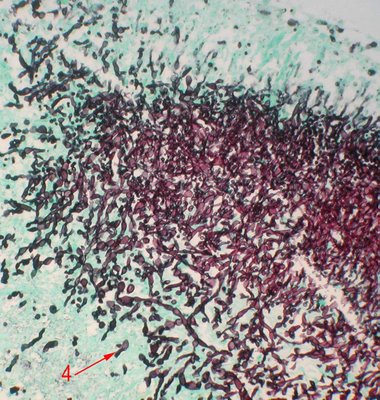
An image of a GMS stained section at the edge of the fungal mass shows budding yeast (red arrow 4). These 3 images depict Candida sp.

Fusarium (red arrow 1) is usually distinguished by its thin hyphae with very rare septae and a right angle branching pattern. Other fungal organisms, including Candida and organisms causing mucormycosis, are visible with routine H&E preparations, although better seen with special stains.
Treatment: Antifungal agents include polyenes, e.g. amphotericin and natamycin, that bind to ergosterol, a component of fungal cell walls, and disrupt the fungal cell wall. Imidazoles, such as ketoconazole, fluconazole interrupt ergosterol synthesis. Fluorinated pyrimidines, such as fluocytosine, work by selective transport mechanism involving a permease into the fungal cell and then blockage of thymidine synthesis.
Candida is often treated with topical amphotericin B (up to 0.3%). Flucytosine may be added and in severe cases oral fluconazole.
Aspergillus filamentary keratitis has been treated with topical amphotericin B, natamycin, as well as oral itraconazole, and ketoconazaole.
Fusarium has been treated with natamycin and ketoconazole.
Mechanical debridement has been used to try to improve penetration of the drug and often is done for diagnostic purposes initially.